Methamphetamine - Wikipedia. This article is about the free base and salts of methamphetamine. For other uses, see Meth (disambiguation). Methamphetamine. INN: Metamfetamine. Clinical data. Pronunciation. Trade names. Desoxyn. AHFS/Drugs. com. Monograph. License data. Pregnancycategory.
US: C (Risk not ruled out)Dependenceliability. Physical: none. Psychological: high. Addictionliability. High. Routes ofadministration. Medical: oral (ingestion), intravenous. Methamphetamine was discovered in 1. It is rarely prescribed due to concerns involving human neurotoxicity and potential for recreational use as an aphrodisiac and euphoriant, among other concerns, as well as the availability of safer substitute drugs with comparable treatment efficacy. 
Dextromethamphetamine is a much stronger CNS stimulant than levomethamphetamine. Both methamphetamine and dextromethamphetamine are illicitly trafficked and sold owing to their potential for recreational use. The highest prevalence of illegal methamphetamine use occurs in parts of Asia, Oceania, and in the United States, where racemic methamphetamine, levomethamphetamine, and dextromethamphetamine are classified as schedule II controlled substances. Levomethamphetamine is available as an over- the- counter (OTC) drug for use as an inhaled nasal decongestant in the United States. While dextromethamphetamine is a more potent drug, racemic methamphetamine is sometimes illicitly produced due to the relative ease of synthesis and limited availability of chemical precursors. In low doses, methamphetamine can elevate mood, increase alertness, concentration and energy in fatigued individuals, reduce appetite and promote (initial) weight loss. At higher doses, it can induce psychosis, breakdown of skeletal muscle, seizures and bleeding in the brain. Chronic high- dose use can precipitate unpredictable and rapid mood swings, prominent delusions and violent behavior. Recreationally, methamphetamine's ability to increase energy has been reported to lift mood and increase sexual desire to such an extent that users are able to engage in sexual activity continuously for several days.  
Heavy recreational use of methamphetamine may lead to a post- acute- withdrawal syndrome, which can persist for months beyond the typical withdrawal period. Unlike amphetamine, methamphetamine is neurotoxic to human midbraindopaminergicneurons. It is related to the other dimethylphenethylamines as a positional isomer of these compounds, which share the common chemical formula: C1. H1. 5N1. Uses. Medical. In the United States, methamphetamine hydrochloride, under the trade name Desoxyn, has been approved by the FDA for treating ADHD and obesity in both adults and children. They suggest the side effect has been exaggerated and stylized to create a stereotype of current users to deter new ones. Following presynaptic dopamine and glutamateco- release by such psychostimulants. Methamphetamine reverses the action of the dopamine transporter (DAT) by activating TAAR1 (not shown). TAAR1 activation also causes some of the dopamine transporters to move into the presynaptic neuron and cease transport (not shown). At VMAT2 (labeled VMAT), methamphetamine causes dopamine efflux (release). Pharmacodynamics. Methamphetamine has been identified as a potent full agonist of trace amine- associated receptor 1 (TAAR1), a G protein- coupled receptor (GPCR) that regulates brain catecholamine systems. At room temperature, the free base of methamphetamine is a clear and colorless liquid with an odor characteristic of geranium leaves. It was used extensively by all branches of the combined Wehrmacht armed forces of the Third Reich, and was popular with Luftwaffe pilots in particular, for its performance- enhancing stimulant effects and to induce extended wakefulness. Side effects were so serious that the army sharply cut back its usage in 1. Suffering from a drug hangover and looking more like a zombie than a great warrior, he had to recover from the side effects. Amphetamine legislation 1. Hearings, Ninety- second Congress, first session, pursuant to S. United States(PDF). Retrieved 1 January 2. We made a decision in January of 1. In humans, the oral bioavailability of methamphetamine is approximately 7. IV) delivery (Ares- Santos et al., 2. 
National Center for Biotechnology Information. Retrieved 3. 1 December 2. United States Food and Drug Administration. Retrieved 3. 0 December 2. Manual of forensic emergency medicine : a guide for clinicians. Sudbury, Mass.: Jones and Bartlett Publishers. Clinical Toxicology (Philadelphia, Pa.). Retrieved 3 January 2. National Center for Biotechnology Information. Retrieved 3. 1 December 2. 
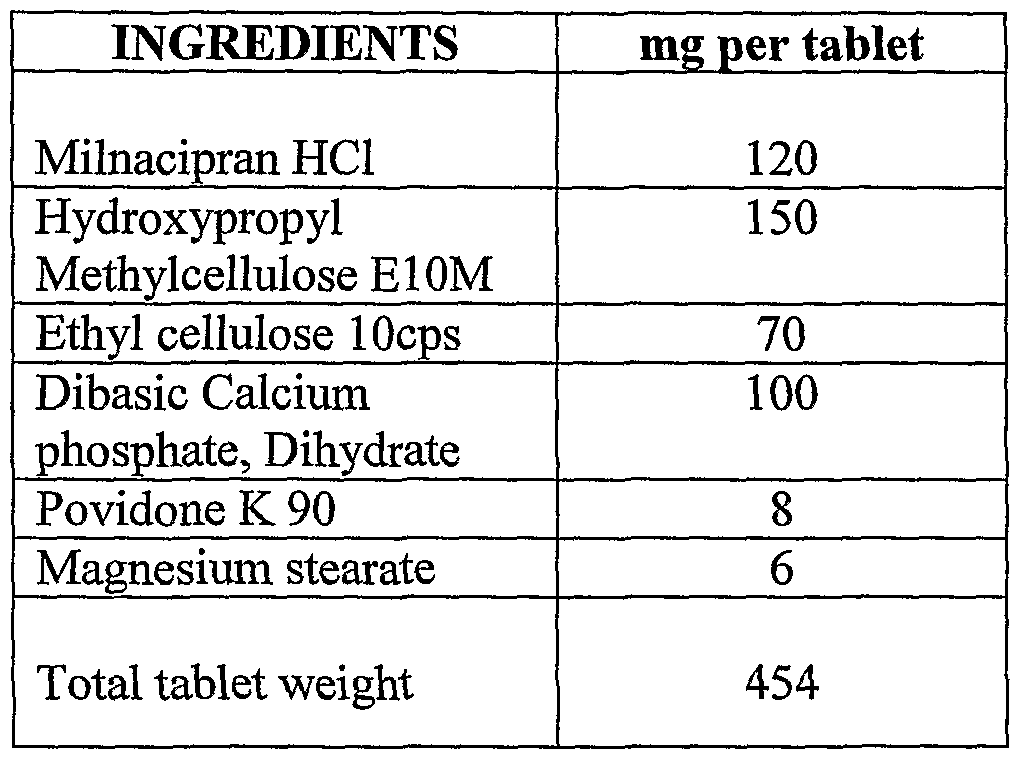
Always combine pharmacologic approaches with nonpharmacologic therapy in the treatment of fibromyalgia (FM), especially stress. Savella (milnacipran) affects certain chemicals in the brain called neurotransmitters. An abnormality in these chemicals is thought to be related to fibromyalgia (a. 
European Monitoring Centre for Drugs and Drug Addiction (EMCDDA). Retrieved 1 September 2. University of Alberta. 
Retrieved 3. 1 December 2. Retrieved 1. 6 January 2. Retrieved 1 January 2. Code of Federal Regulations Title 2. Subchapter D – Drugs for human use. United States Food and Drug Administration. Retrieved 7 March 2. Topical nasal decongestants - -(i) For products containing levmetamfetamine identified in 3. The product delivers in each 8. National Center for Biotechnology Information. 
Retrieved 2 January 2. National Geographic Channel. Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: Mc. Graw- Hill Medical. ISBN 9. 78- 0- 0. 
Unlike cocaine and amphetamine, methamphetamine is directly toxic to midbrain dopamine neurons. Neuroimaging studies have revealed that METH can indeed cause neurodegenerative changes in the brains of human addicts (Aron and Paulus, 2. Chang et al., 2. 00. These abnormalities include persistent decreases in the levels of dopamine transporters (DAT) in the orbitofrontal cortex, dorsolateral prefrontal cortex, and the caudate- putamen (Mc. Cann et al., 1. 99. Sekine et al., 2. Volkow et al., 2. The density of serotonin transporters (5- HTT) is also decreased in the midbrain, caudate, putamen, hypothalamus, thalamus, the orbitofrontal, temporal, and cingulate cortices of METH- dependent individuals (Sekine et al., 2. Neuropsychological studies have detected deficits in attention, working memory, and decision- making in chronic METH addicts .. There is compelling evidence that the negative neuropsychiatric consequences of METH abuse are due, at least in part, to drug- induced neuropathological changes in the brains of these METH- exposed individuals .. Structural magnetic resonance imaging (MRI) studies in METH addicts have revealed substantial morphological changes in their brains. These include loss of gray matter in the cingulate, limbic and paralimbic cortices, significant shrinkage of hippocampi, and hypertrophy of white matter (Thompson et al., 2. In addition, the brains of METH abusers show evidence of hyperintensities in white matter (Bae et al., 2. Ernst et al., 2. 00. N- acetylaspartate (Ernst et al., 2. Sung et al., 2. 00. Sekine et al., 2. Chang et al., 2. 00. Ernst et al., 2. 00. Sung et al., 2. 00. Yen et al., 1. 99. Elevated choline levels, which are indicative of increased cellular membrane synthesis and turnover are also evident in the frontal gray matter of METH abusers (Ernst et al., 2. Salo et al., 2. 00. Taylor et al., 2. United States Food and Drug Administration. Retrieved 6 January 2. Neuropsychopharmacology. Retrieved 6 March 2. Goldfrank's toxicologic emergencies (9th ed.). New York: Mc. Graw- Hill Medical. In Brunton LL, Chabner BA, Knollmann BC. Goodman & Gilman's Pharmacological Basis of Therapeutics (1. New York: Mc. Graw- Hill. ISBN 9. 78- 0- 0. The Southeast Asian Journal of Tropical Medicine and Public Health. American Family Physician. American Dental Association. Archived from the original on June 2. Retrieved December 2. Neuropsychopharmacology. Retrieved January 2. Archived from the original(PDF) on August 2. Retrieved January 2. Merck Manual for Health Care Professionals. Glia (including astrocytes, microglia, and oligodendrocytes), which constitute the majority of cells in the brain, have many of the same receptors as neurons, secrete neurotransmitters and neurotrophic and neuroinflammatory factors, control clearance of neurotransmitters from synaptic clefts, and are intimately involved in synaptic plasticity. Despite their prevalence and spectrum of functions, appreciation of their potential general importance has been elusive since their identification in the mid- 1. This development of appreciation has been nurtured by the growing awareness that drugs of abuse, including the psychostimulants, affect glial activity, and glial activity, in turn, has been found to modulate the effects of the psychostimulants ^Loftis JM, Janowsky A (2. Collectively, these pathological processes contribute to neurotoxicity (e. BBB permeability, inflammation, neuronal degeneration, cell death) and neuropsychiatric impairments (e. They are present in the organs that mediate the actions of METH (e. In the brain, METH acts primarily on the dopaminergic system to cause acute locomotor stimulant, subchronic sensitized, and neurotoxic effects. Repeated administration or self administration of METH has been shown to upregulate . The Journal of Pharmacology and Experimental Therapeutics. TAAR1 overexpression significantly decreased EAAT- 2 levels and glutamate clearance .. METH treatment activated TAAR1 leading to intracellular c. AMP in human astrocytes and modulated glutamate clearance abilities. Furthermore, molecular alterations in astrocyte TAAR1 levels correspond to changes in astrocyte EAAT- 2 levels and function. TAAR1 is largely located in the intracellular compartments both in neurons (Miller, 2. Cisneros and Ghorpade, 2. Grandy, 2. 00. 7) ^ abcd. Rodvelt KR, Miller DK (September 2. In Olson KR, Anderson IB, Benowitz NL, Blanc PD, Kearney TE, Kim- Katz SY, Wu AH. Poisoning & Drug Overdose (6th ed.). New York: Mc. Graw- Hill Medical. ISBN 9. 78- 0- 0. Retrieved 1. 1 June 2. Shoptaw SJ, Ali R, eds. Cochrane Database Syst. Sertraline - Wikipedia. For the electronic music band, see Lustral (band). Sertraline. Clinical data. Trade names. Zoloft and others. It was introduced to the market by Pfizer in 1. Sertraline is primarily prescribed for major depressive disorder in adult outpatients as well as obsessive- compulsive disorder, panic disorder, and social anxiety disorder, in both adults and children. In 2. 01. 3, it was the most prescribed antidepressant and second most prescribed psychiatric medication (after alprazolam) on the U. S. It is similar in tolerability profile to other SSRIs, with the types of adverse events usually including diarrhea, nausea, and sexual dysfunction. The incidence of diarrhea was higher with sertraline—especially when prescribed at higher doses—in comparison to other SSRIs. A 1. 99. 8 review suggested that, due to its pharmacology, sertraline may be more efficacious than other SSRIs and equal to TCAs for the treatment of melancholic depression. Reboxetine was significantly worse. Sertraline had much lower rates of adverse effects than these TCAs, with the exception of nausea, which occurred more frequently with sertraline. In addition, sertraline appeared to be more effective than fluoxetine or nortriptyline in the older- than- 7. With respect to safety duloxetine and venlafaxine increased worsened dizziness, however not much safety data was reported. The response rate was independent of the dose. In addition to decreasing the frequency of panic attacks by about 8. The patients rated as . The authors of the study argued that the improvement achieved with sertraline is different and of a better quality than the improvement achieved with placebo. Significant improvement was observed in 5. The improvement began during the first week of treatment, and in addition to mood, irritability, and anxiety, improvement was reflected in better family functioning, social activity and general quality of life. Work functioning and physical symptoms, such as swelling, bloating and breast tenderness, were less responsive to sertraline. Of the sertraline group, 4. This result compares favorably with placebo, where, according to the literature, 3–6% of patients gained more than 7% of their initial weight. The large weight gain was observed only among female members of the sertraline group; the significance of this finding is unclear because of the small size of the group. Divided attention was improved and verbal memory under interference conditions decreased marginally. Because of the large number of measures taken, it is possible that these changes were still due to chance. The observed frequency of sexual side effects depends greatly on whether they are reported by patients spontaneously, as in the manufacturer's trials, or actively solicited by the physicians. There have been several double- blind studies of sexual side effects comparing sertraline with placebo or other antidepressants. The mood improvement resulting from the treatment with sertraline sometimes counteracted these side effects, so that sexual desire and overall satisfaction with sex stayed the same as before the sertraline treatment. However, under the action of placebo the desire and satisfaction slightly improved. This warning is based on statistical analyses conducted by two independent groups of FDA experts that found a twofold increase of suicidal ideation and behavior in children and adolescents, and a 1. For the above analysis, the FDA combined the results of 2. Considered separately, sertraline use in adults decreased the odds of suicidal behavior with a marginal statistical significance by 3. The authors of the FDA analysis note that . Plasma, serum or blood concentrations of sertraline and norsertraline, its major active metabolite, may be measured to confirm a diagnosis of poisoning in hospitalized patients or to aid in the medicolegal investigation of fatalities. As a result, more than half of breast- fed babies receive less than 2 mg/day of sertraline and desmethylsertraline combined, and in most cases these substances are undetectable in their blood. The symptoms can include flu- like symptoms and disturbances in sleep, senses, movement, mood, and thinking. In most cases symptoms are mild, short- lived, and resolve without treatment. More severe cases are often successfully treated by temporary reintroduction of the drug with a slower tapering off rate. In a placebo- controlled study in which sertraline was co- administered with lithium, 3. Sertraline concentrate contains alcohol, and is therefore contraindicated with disulfiram (Antabuse). The prescribing information recommends that treatment of the elderly and patients with liver impairment . However, this is not considered a tight binding, and this action is only 1. In the blood, it is 9. Its half- life in the body is 1. It appeared unlikely that inhibition of any single isoform could cause clinically significant changes in sertraline pharmacokinetics. These conclusions have not been verified in human studies. Sertraline deaminated is O- 2. Development of tametraline was soon stopped because of undesired stimulant effects observed in animals. A few years later, in 1. Kenneth Koe, after comparing the structural features of a variety of reuptake inhibitors, became interested in the tametraline series. He asked another Pfizer chemist, Willard Welch, to synthesize some previously unexplored tametraline derivatives. Welch generated a number of potent norepinephrine and triple reuptake inhibitors, but to the surprise of the scientists, one representative of the generally inactive cis- analogs was a serotonin reuptake inhibitor. Welch then prepared stereoisomers of this compound, which were tested in vivo by animal behavioral scientist Albert Weissman. The most potent and selective (+)- isomer was taken into further development and eventually named sertraline. Weissman and Koe recalled that the group did not set up to produce an antidepressant of the SSRI type—in that sense their inquiry was not . According to Welch, they worked outside the mainstream at Pfizer, and even . The group had to overcome initial bureaucratic reluctance to pursue sertraline development, as Pfizer was considering licensing an antidepressant candidate from another company. Food and Drug Administration (FDA) in 1. Psychopharmacological Drugs Advisory Committee; it had already become available in the United Kingdom the previous year. In 2. 00. 3, the UK Medicines and Healthcare products Regulatory Agency issued a guidance that, apart from fluoxetine (Prozac), SSRIs are not suitable for the treatment of depression in patients under 1. In 2. 00. 7, labeling was again changed to add a warning regarding suicidal behavior in young adults ages 1. Goodman and Gilman’s The Pharmacological Basis of Therapeutics, Twelfth Edition. Mc. Graw Hill Professional; 2. Sertraline Page accessed 1. May 2. 01. 5^ abcd. Obach RS, Cox LM, Tremaine LM (2. Retrieved 3 April 2. International Clinical Psychopharmacology. The American Society of Health- System Pharmacists. Retrieved 3 April 2. International Clinical Psychopharmacology. J Clin Psychopharmacol. Current Medical Research and Opinion. K.; Khosla, P; Deswal, R. Int Clin Psychopharmacol. Psychotherapy and psychosomatics. Health Technol Assess. Suppl B): S9. 9–1. Lay summary – The Washington Post (2. January 2. 00. 9). Canadian Journal of Psychiatry. The Journal of Clinical Psychiatry. The Journal of Clinical Psychiatry. The Cochrane database of systematic reviews (4): CD0. A meta- analysis of pharmacotherapy trials in pediatric obsessive- compulsive disorder. The American Journal of Psychiatry. The Journal of Clinical Psychiatry. Retrieved 2. 8 July 2. The Journal of Clinical Psychiatry. Arch Womens Ment Health. Int Clin Psychopharmacol. The New England Journal of Medicine. BMJ clinical evidence. The Cochrane database of systematic reviews. Current clinical pharmacology. The journal of sexual medicine. Expert opinion on pharmacotherapy. The Maudsley prescribing guidelines in psychiatry. West Sussex: Wiley- Blackwell. ISBN 9. 78- 0- 4. Fluoxetine Hydrochloride. Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 2. 7 November 2. Wolters Kluwer Health. Retrieved 2. 7 November 2. The Journal of Clinical Psychiatry. The Journal of Clinical Psychiatry. Psychopharmacology (Berl.). Psychopharmacology (Berl.). J Child Adolesc Psychopharmacol. Journal of psychopharmacology (Oxford, England). The Journal of Clinical Psychiatry. Clinical Therapeutics. Retrieved 1. 1 July 2. Overview for December 1. Meeting of Psychopharmacologic Drugs Advisory Committee (PDAC). Retrieved 1. 1 July 2. FDA DOCKET 2. 00. N- 0. 41. 4. Retrieved 1. July 2. 00. 8. Retrieved 1. July 2. 00. 8. The British Journal of Psychiatry. Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, California: Biomedical Publications. Journal of Medical Toxicology. The American Journal of Psychiatry. The Journal of Clinical Psychiatry. The American Journal of Psychiatry. Journal of Clinical Psychopharmacology. Journal of Clinical Psychopharmacology. Journal of Clinical Pharmacology. Clinical Pharmacokinetics. Journal of Clinical Psychopharmacology. The American Journal on Addictions. ADHD comorbidities: handbook for ADHD complications in children and adults. American Psychiatric Pub. ISBN 9. 78- 1- 5. Journal of Clinical Psychopharmacology. Journal of clinical pharmacology. Journal of Clinical Psychopharmacology. Biological Psychiatry. The American Journal of Psychiatry. Central Nervous System Agents in Medicinal Chemistry. Life Sci (5. 2): 1. Delhi Psychiatry Journal. Pharmacotherapy of Depression. Springer. Link (2nd ed.). New York, NY: Humana Press. ISBN 9. 78- 1- 6. Advances in Medicinal Chemistry. Advances in Medicinal Chemistry. ISBN 9. 78- 1- 5. A Class of Compounds with a Novel Profile of Central Nervous System Activity. Journal of Medicinal Chemistry. Chemical & Engineering News. The Antidepressant Era.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2017
Categories |
 RSS Feed
RSS Feed
